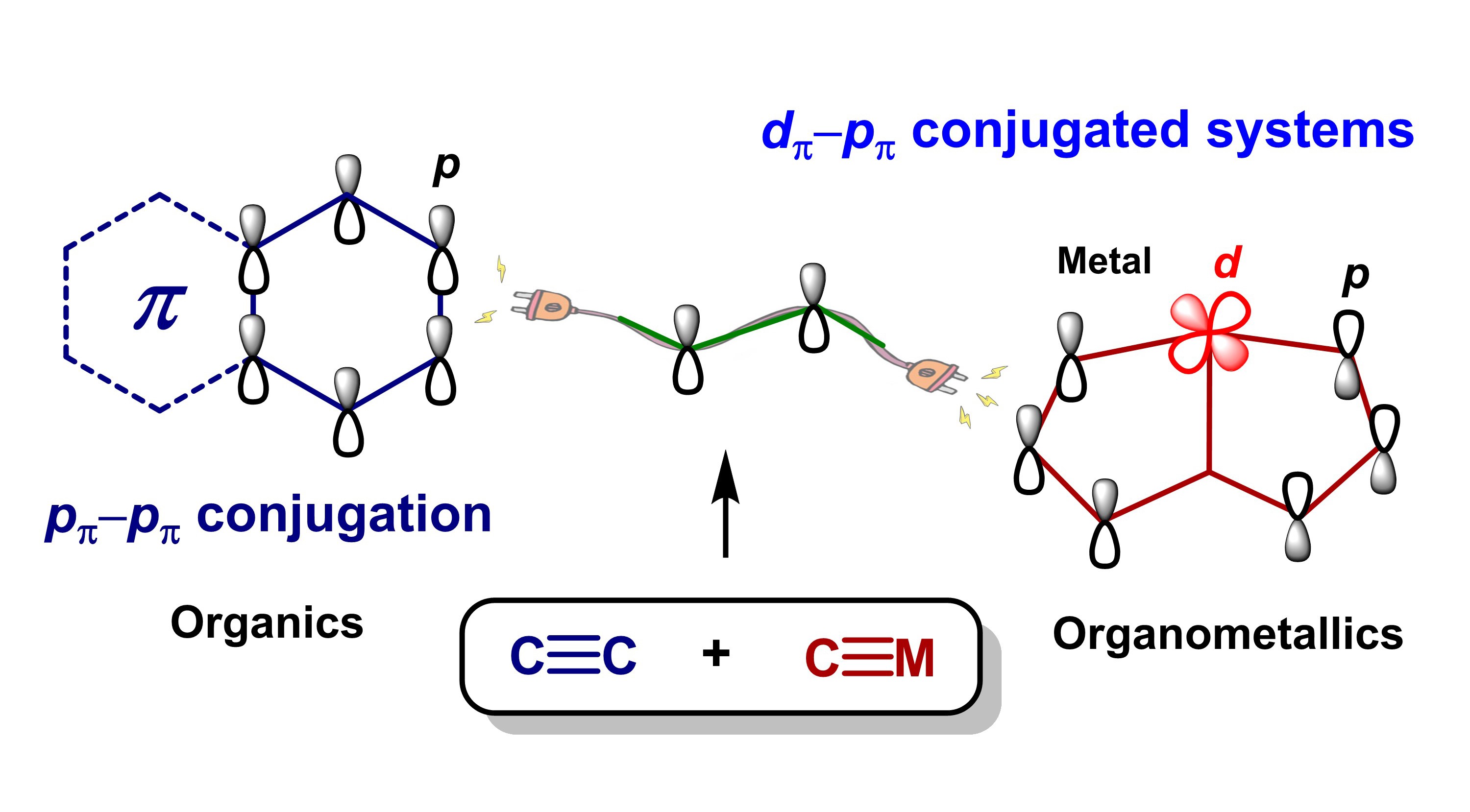
The metal-carbon triple bonds and carbon-carbon triple bonds are both highly unsaturated bonds. As a result, their reactions tend to afford cycloaddition intermediates or products. Herein, we report a reaction of M≡C and C≡C bonds that affords acyclic addition products. These newly discovered reactions are highly efficient, regio- and stereospecific, with good functional group tolerance, and are robust under air at room temperature. The isotope labeling NMR experiments and theoretical calculations reveal the reaction mechanism. Employing these reactions, functionalized dπ-pπ conjugated systems can be easily constructed and modified. The resulting dπ-pπ conjugated systems were found to be good electron transport layer materials in organic solar cells, with power conversion efficiency up to 16.28% based on the PM6: Y6 non-fullerene system. This work provides a facile, efficient methodology for the preparation of dπ-pπ conjugated systems for use in functional materials.
URL link for article: https://www.nature.com/articles/s41467-020-18498-2